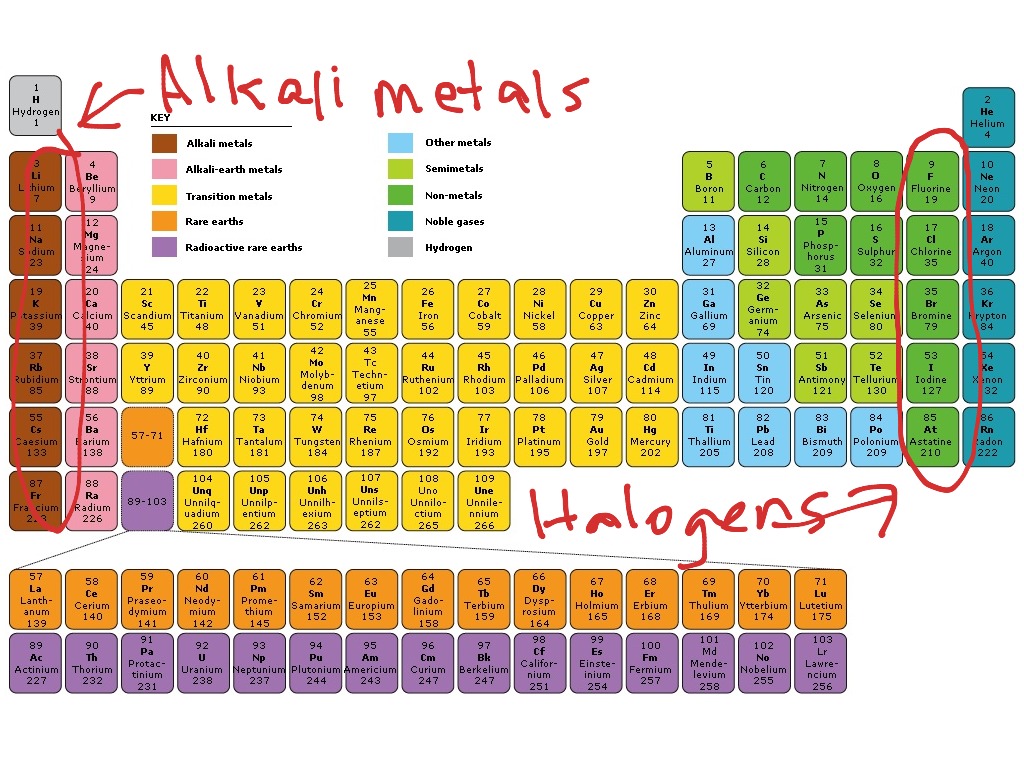
Most of these compounds are ionic in nature. Potassium + chlorine → potassium chlorideĪll alkalis manifest themselves as a white solid in their compound form that is capable of being dissolved in water. Following chemical equations demonstrate some of the ways in which they react: How Do Alkali Metals React Otherwise?Īs alkali metals are the most reactive group of metals in the periodic table, each of these metals is capable of reacting with different elements to produce different results. Potassium + water → potassium hydroxide + hydrogenĪll elements in a particular group react in an analogous manner. Sodium + water → sodium hydroxide + hydrogenĢ Na(s) + 2 H 2O(l) → 2 NaOH (aq) + H 2(g) Lithium + water → lithium hydroxide + hydrogenĢ Li(s) + 2 H 2O(l) → 2 LiOH (aq) + H 2(g) The following chemical equations demonstrate how various metals react with water: Alkaline metals when reacting with water produce an alkaline solution, along with the release of hydrogen gas. Alkali Metals Reactivity with WaterĪlkali metals derive their classification because of the results of their reaction with water. As a conductor of heat and electricity, they are excellent. Upon being cut into two halves, we can observe that their surface is as shiny as any other metal but even after they are stored in oil, they undergo tarnishing. Therefore, if we decide to cut these metals, we will be able to do so, without much trouble. When it comes to the density of the metal, group one elements display a very low level of density of up to 1 gcm -3 which means that they can easily float on the surface of the water. The melting points of these elements are quite low, which is 180° Celsius in the case of Lithium, while it is 39° Celsius in the case of Rubidium. In order to prevent the elements from coming in contact with oxygen, they are stored in jars that contain oil. Caesium is the most metallic element in the group. As we move down the group, the atomic radius increases. They have the least nuclear charge in their respective periods. They have a strong tendency to donate their valence electron in the last shell to form strong ionic bonds. The general electronic configuration of elements of group 1 is ns 1.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
